
Innovative software with customised support
for clinical committee management
Streamline processes without compromising quality: Clinical Adjudication, Protocol Deviations, Data and Safety Monitoring Boards, Data Monitoring Committees, Steering Committees and more.
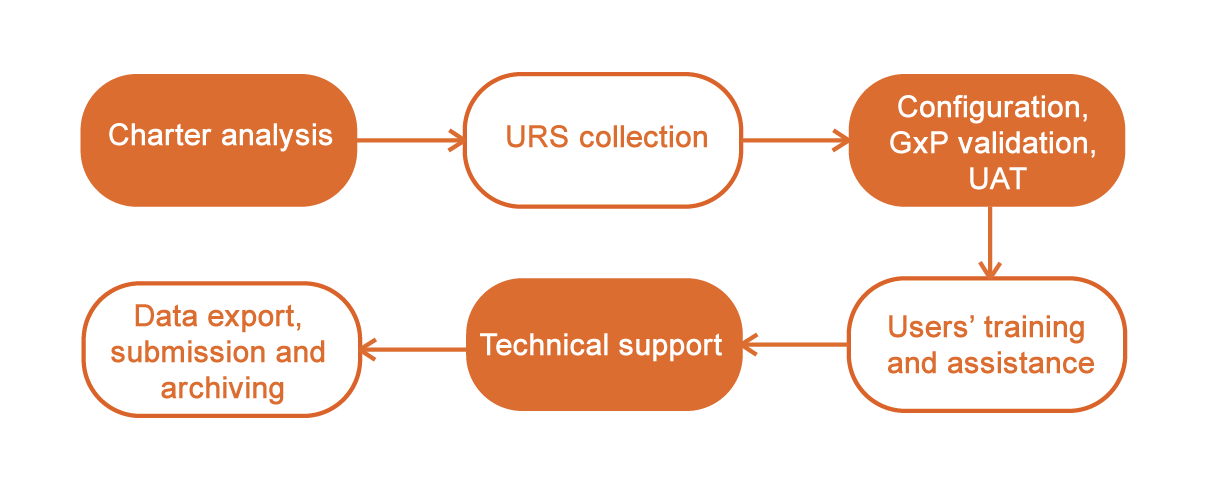
Our IT teams configure the software to match exactly your charter. From User Requirements Specifications to study archival through GxP validation, we support you throughout your clinical committee management process.
.

Equipped with automation features, smart tools and hands-on task management features, eReconciliation® allows data management, clinical and safety teams to perform reconciliation all along the study duration with minimum burden while keeping a full audit-trail as requested during regulatory inspections.

Ethical’s software and business processes are built to meet globally recognized industry standards and regulatory requirements. These include the ISO/IEC 27001 for Information Security Management (ISMS), Good Clinical Practice (GCP), US FDA 21 CFR Part 11, EU GMP Vol. 4 Annex 11, the EU General Data Protection Regulation (GDPR), and GAMP 5 Documentation.
Ethical software design and the associated services are based on a hardware/software and organizational structure that ensures compliance with the ISO/IEC 27001 Information Security Management System (ISMS) requirements and security controls. Ethical's parent company GM Servizi is audited yearly by an authorized ISO 27001Certification Authority.
Ethical’s softwares, systems, and services comply with all regulations that apply to EA: GxP, GAMP 5, US 21 CFR Part 11, EU GMP Vol. 4 Annex 11.
Ethical provides the full GAMP 5 validation documentation package and support.
Ethical GmbH is committed to ensuring the security and protection of the personal information and to provide a compliant and consistent approach to data protection. We have a robust and effective data protection program in place which complies with European Union General Data Protection Regulation (EU-GDPR)

eAdjudication
software solution
service description